Prospera™ Lung Transplant Assessment – the next generation
- A single blood test to assess all transplant rejection types
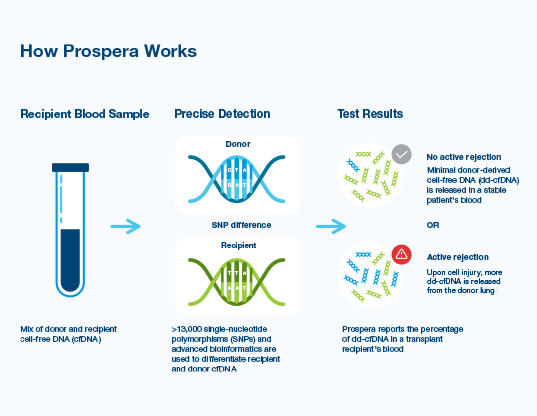
- The blood sample contains a mixture of donor DNA (shed from the donor organ) and patient DNA
- Prospera™ differentiates between donor DNA and patient DNA using differences in single-nucleotide polymorphisms
- Prospera™ reports the percentage of dd-cfDNA in the patient’s blood, a predictor of acute rejection
Experts in cell-free DNA
Optimized for transplant
- Developed by Natera, a leader in cfDNA with a trusted legacy in fetal monitoring, oncology and organ health
- Demonstrated in over 10 million tests1
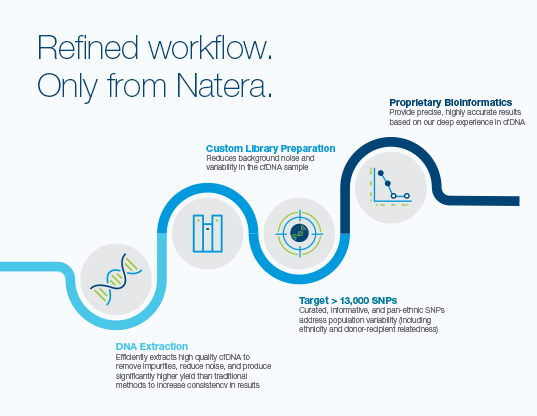
- Utilizes over 13,000 pan-ethnic SNPs and advanced bioinformatics2
- Optimized to be a precise cfDNA tool for early, clinically meaningful rejection assessment.3
Precision optimized for enhanced performance
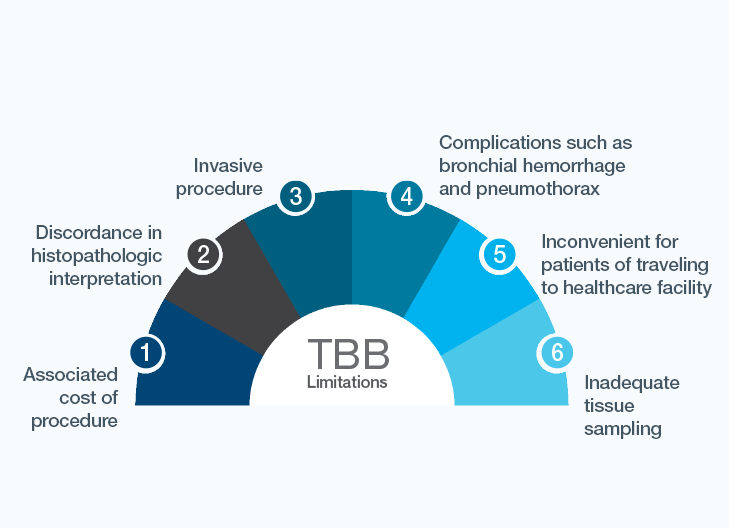
Reduce the number of surveillance biopsies by monitoring with Prospera™ Lung
-
A noninvasive surveillance test that allows you to determine when a biopsy should be performed on a clinically stable patient
-
Prospera™ Lung is an intuitive biomarker for allograft injury with excellent performance
-
Better science for better care
-
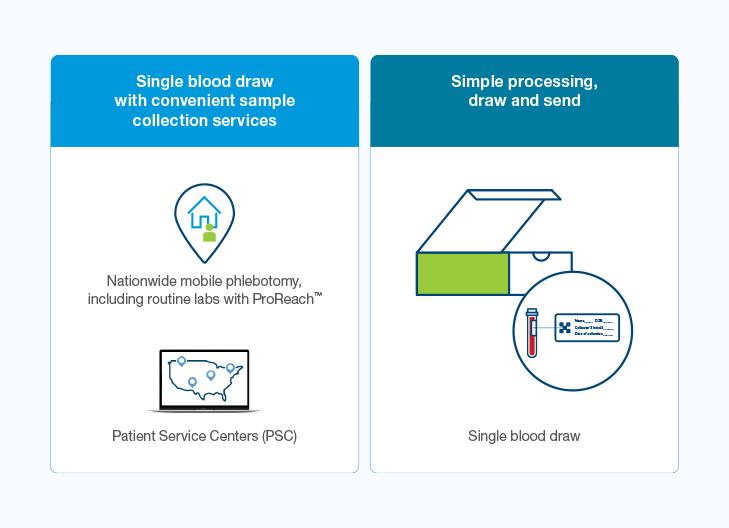
Easy-to-use with a single blood draw that requires no special processing

New Publication! Single Lung Allograft Monitoring study (SLAM)4
- SLAM is the largest study of commercial dd-cfDNA tests in single lung transplant (SLT)
- The SLAM study is a multi-center, retrospective analysis from 6 academic lung transplant centers in the US.
- The aim of the SLAM study is to demonstrate Prospera™ Lung performance in assessing transplant rejection in the single lung transplant (SLT) population.
SLAM study establishes Prospera™ Lung as an accurate, noninvasive transplant assessment for Single Lung Transplantation
Patients First, Partners Always
Natera’s suite of solutions allows for streamlined process integration for your center

Total EMRSync
A complete, secure bi-directional data and workflow interface with Epic and Cerner systems
- Allows for easy ordering and delivery of results

ProsperaLink Program
A concierge team of clinical experts including a medical science liaison, nurse coordinator, & patient coordinator to help patients stay updated on blood draws, compliance plans and results.

Financial Support Program
- Natera welcomes all insurances, and our goal is to make the process easy and transparent for patients
- In the rare event your patient has financial responsibility, Natera offers flexible financial assistance programs and will work closely with your patient to ensure there is no hardship on them or their family.
Find out more about Prospera™ for lung transplant recipients
References
1Natera Inc. Natera validation data: manuscript submitted. Data on file.
2Sigdel TK, Archila FA, Constantin T, et al. Optimizing detection of kidney transplant injury by assessment of donor-derived cell-free DNA via massively multiplex PCR. J Clin Med. 2018 (per published article);8(1):19 doi:10.3390/jcm8010019
3Rosenheck JP, Ross DJ, Botros, M, et al. Clinical Validation of a Plasma Donor-derived Cell-free DNA Assay to Detect Allograft Rejection and Injury in Lung Transplant. Transplantation Direct. 2022;8(4) doi:10.1097/txd.0000000000001317
4Arunachalam A, Anjum F, Rosenheck JP, et al. Donor-derived Cell-free DNA is a Valuable Monitoring Tool after Single Lung Transplantation: Multi-center Analysis. JHLT Open. 2024. doi:https://doi.org/10.1016/j.jhlto.2024.100155